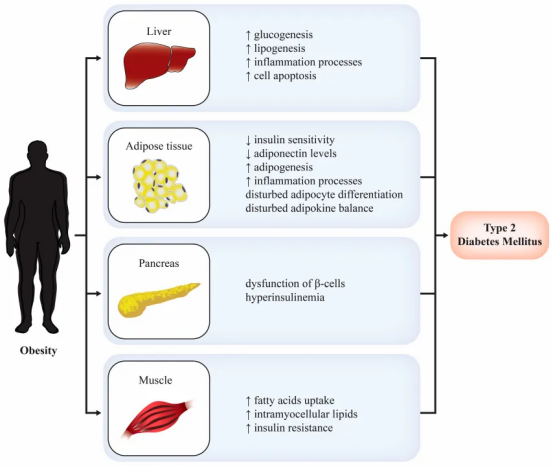
Currently, overweight and obesity have become one of the most serious global health challenges. According to data from the International Diabetes Federation (IDF), the number of adults living with diabetes worldwide reached 588.7 million in 2024. It is projected that by 2050, this number will increase to 852.5 million, accounting for 13% of the global population.
GLP-1 receptor agonists have become a major class in the markets for glycemic control and weight management, with products such as semaglutide and tirzepatide achieving steadily increasing annual sales. At the same time, multiple targets closely related to mechanisms such as energy metabolism, insulin secretion, and appetite regulation also play critical roles in drug development, collectively contributing to a more comprehensive understanding of this field.
01 GLP-1R: A Core Therapeutic Target
GLP-1R is a G protein–coupled receptor expressed in pancreatic cells and neurons in the brain. Upon binding with GLP-1, it activates the cAMP/PKA signaling pathway, promoting insulin gene transcription and secretion. This target remains central to the development of therapies for diabetes and weight management and is one of the most well-validated directions in the current market.
02 GIPR: A Key Regulator of the Incretin Effect
GIPR is the receptor for GIP and belongs to the seven-transmembrane G protein–coupled receptor family. The interaction between GIP and GIPR directly regulates insulin secretion, blood glucose levels, and lipid metabolism. In the context of glycemic control and weight management, GIPR works synergistically with GLP-1R, and dual agonists (such as tirzepatide) have demonstrated superior efficacy in both glucose and weight control in clinical settings.
03 GCGR: Balancing Glycemic Control and Energy Metabolism
GCGR is primarily expressed in pancreatic β cells and hepatocytes. Its activation promotes insulin secretion and plays a role in regulating hepatic gluconeogenesis. Studies suggest that appropriate modulation of GCGR activity can help reduce blood glucose levels and, when combined with GLP-1R activity, offers complementary benefits in weight management and metabolic improvement.
04 FGF-21: A Liver-Derived Metabolic Regulator
FGF21 is a hormone produced by the liver that enhances energy expenditure, improves insulin sensitivity, and promotes brown adipose tissue activation. These effects complement the mechanisms of GLP-1-based therapies, making it an important target in the development of treatments for obesity, nonalcoholic steatohepatitis (NASH), and type 2 diabetes.
05 Amylin: Regulating Appetite at the Source
Amylin is a satiety peptide co-secreted with insulin. By activating amylin receptors (AMYRs), it induces satiety signals, reduces food intake, and supports energy metabolism while decreasing adipose tissue mass. In the context of glycemic control and weight management, amylin analogs (such as pramlintide) have been used as adjuncts to insulin therapy, and next-generation dual-target therapies are under rapid development.
06 ActRII: Improving Body Composition
ActRII (activin type II receptor) is a member of the TGF-β receptor family. Overactivation of this pathway is associated with muscle atrophy and fat accumulation. Targeting ActRII enables simultaneous fat reduction and muscle gain, addressing an important limitation observed with long-term use of GLP-1–based therapies.
Drug Development Progress
As the GLP-1 market continues to expand, oral weight-loss therapies have become a key area of competition. By 2030, oral drugs are expected to account for approximately 20% of the GLP-1 weight-loss market, with the total market size projected to reach around $80 billion.
In the oral GLP-1 space, the primary competition is between Novo Nordisk and Eli Lilly. The former is expected to lead with its oral version of Wegovy, followed closely by orforglipron from Eli Lilly.
Novo Nordisk: Its oral semaglutide (Wegovy® tablet) utilizes SNAC absorption enhancer technology to protect the peptide from degradation. As of early April 2026, cumulative prescriptions have exceeded 600,000. However, it must be taken on an empty stomach in the morning, with no food or water allowed for 30 minutes after dosing.
Eli Lilly: The company has developed orforglipron, an oral small-molecule GLP-1 receptor agonist that can be taken with meals, offering greater convenience. Its weight-loss efficacy (16.6%) is comparable to Novo Nordisk’s product, with similar pricing and the advantage of sufficient supply capacity.
Beyond the direct competition between these two companies, research and development in the oral weight-loss drug field continues to advance.
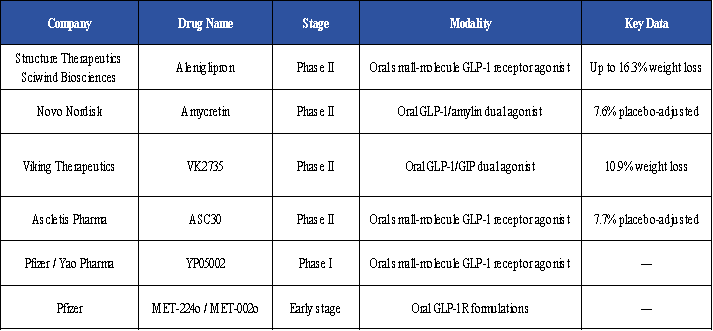
From Single Targets to Multi-Target Synergy
GLP-1R remains the foundational target for glycemic control and weight management. However, a single-target approach is insufficient to address the diverse needs of all patient populations. Targets such as GIPR, GCGR, FGF21, Amylin, and ActRII contribute through different mechanisms, including insulin secretion, energy expenditure, appetite regulation, and body composition improvement, thereby complementing GLP-1-based therapies.
Over the next 3–5 years, as clinical data from dual-target, multi-target, and oral formulations continue to emerge, the field of metabolic disease treatment is expected to shift toward more precise and personalized combination strategies.
About Genomeditech
Genomeditech focuses on cutting-edge research in metabolic diseases, offering an innovative product portfolio covering key targets such as GLP-1R/GCGR/GIPR, ActRII, AMY/CALCR, and FGF21, along with an efficient service system. Aligned with emerging trends in GLP-1R-, GIPR-, and GCGR-based multi-agonist therapies, Genomeditech provides global pharmaceutical companies with integrated, end-to-end solutions from early-stage research to clinical development.
Target-related cell lines:
Engineered with validated receptor expression and functional activity, these cell lines are applicable for studies on GLP-1R, GIPR, and GCGR signaling pathways, cell-based drug activity assays and screening, as well as CMC quality control and release testing.
High-activity recombinant proteins:
These proteins are suitable for antibody immunization and screening against GCGR, ligand–receptor binding and functional studies, and in vitro binding assays (e.g., ELISA, SPR, BLI). They provide reliable biological materials for mechanism research and drug development targeting GLP-1R/GCGR and related metabolic pathways.
High-specificity antibodies:
Validated human IgG monoclonal antibodies are available for applications including flow cytometry, immunohistochemistry, Western blot, and ELISA, delivering accurate and reproducible data for target validation and drug discovery.
Reference
International Diabetes Federation. (2025). IDF Diabetes Atlas (11th ed.). https://international-diabetes-federation.s3.eu-west-1.amazonaws.com/media/uploads/sites/3/2025/10/IDF_Diabetes_Atlas_11th_Edition_2025_WEB.pdf
Structure Therapeutics. (2025). Structure Therapeutics reports positive topline data from Phase 2 study. https://ir.structuretx.com/news-releases/news-release-details/structure-therapeutics-reports-positive-topline-data-phase-2
Eli Lilly and Company. (2025, September 16). Lilly’s oral GLP-1 orforglipron demonstrated meaningful weight loss and cardiometabolic improvements in Phase 3 study. https://investor.lilly.com/news-releases/news-release-details/lillys-oral-glp-1-orforglipron-showed-compelling-efficacy-and
Viking Therapeutics. (2025, August 19). Viking Therapeutics announces positive top-line results from Phase 2 VENTURE oral dosing trial of VK2735 tablet formulation in patients with obesity. https://ir.vikingtherapeutics.com/2025-08-19-Viking-Therapeutics-Announces-Positive-Top-Line-Results-from-Phase-2-VENTURE-Oral-Dosing-Trial-of-VK2735-Tablet-Formulation-in-Patients-with-Obesity
Ascletis Pharma Inc. (2026). [Company presentation/report]. https://www.ascletis.com/data/upload/admin/20260112/69644fd640af4.pdf
Pfizer Inc. (2025). Pfizer enters exclusive collaboration and license agreement. https://www.pfizer.com/news/press-release/press-release-detail/pfizer-enters-exclusive-collaboration-and-license-agreement
Pfizer Inc. (2025). Pfizer to acquire Metsera and its next-generation obesity portfolio. https://www.pfizer.com/news/press-release/press-release-detail/pfizer-acquire-metsera-and-its-next-generation-obesity